
Users have therefore been calling for an in-line testing capability in carrying out VDA 19 compliant residual contamination monitoring. This process, being decoupled as it were from the manufacturing environment, involves a time delay in the quality control loop which harbors the risk that already assembled parts may have to be re-cleaned or parts already shipped may need to be recalled at a high expenditure of time and cost. Here, particulate contaminants are first extracted from the part by means of a fluid before being collected on a filter and subsequently analyzed to customer specifications. In accordance with VDA 19 or ISO 16232, residual contamination analyses are then carried out in a separate laboratory. Quite often the particles specified as critical are found on interior part surfaces, e.g., inside the oil passages of a crankcase, so that tests relying on direct measurement methods are not feasible. 
However, checking and documenting this so-called technical cleanliness presents a challenge to OEM and tiered suppliers alike. 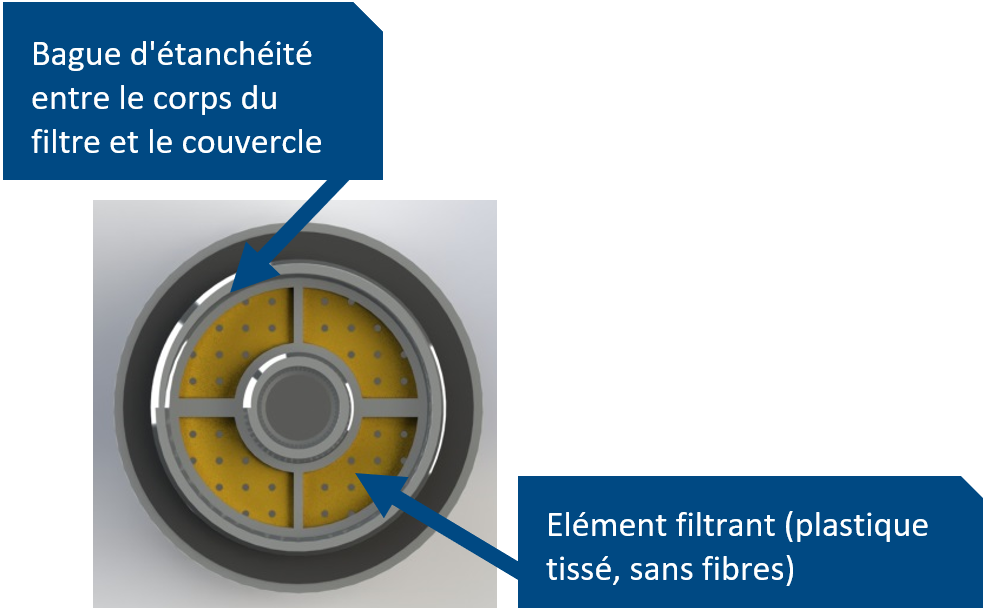
Particulate cleanliness constitutes one of the most important quality factors today, both in the automotive industry and in numerous other manufacturing sectors. Thanks to this technology, results are obtained much faster and process reliability increases significantly. With its new EcoCLab system, Dürr Ecoclean now offers an automated in-line capability for inspecting and documenting particulate cleanliness in compliance with VDA 19. That is how we can find a solution for your controlled area that matches your requirements perfectly.One major drawback of cleanliness testing used to lie in the associated time-consuming and costly laboratory methods. Here at PETEK, we are always pleased to meet you for a consulting session. As experienced cleanroom experts, we here at PETEK are always pleased to assist you. If you have any questions about which requirements apply to your sector, you are best to contact us directly. 
a choice of various equipment options for the controlled area.inexpensive controlled area, produced to your requirements.This greatly reduces the construction cost of a cleanroom.īased on these standards, we would be pleased to provide you with a made-to-measure solution: This means that the purity requirements are not as stringent as they are in, for example, medical cleanrooms. However, sterile rooms must be classified in the lower classes of cleanroom. In terms of its basic structure, a controlled area is no different from other cleanrooms (such as Technical Cleanrooms) in which the number of airborne particles is reduced by filtering the intake air. In sterile rooms (and in grey rooms), product contamination is reduced to a defined extent. For example, VDA 19 and ISO 16232 stipulate the residual contamination requirements in relation to technical cleanliness (TecSa) and purity level (SaS) that need to be satisfied.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
